This publication presents the 12-month follow-up of the NIHP2019 trial, the first multicentre, randomised controlled study evaluating hypothermic oxygenated machine perfusion (HOPE) for donor heart preservation in adult heart transplantation.
In this prespecified secondary analysis, 204 transplanted patients from 15 European centres were followed for 12 months after transplantation. The key secondary endpoint of the trial consisted of a composite endpoint occurring within 12 months after transplant including; any cause of death, primary graft dysfunction (PGD) (moderate, severe, and right ventricular), graft failure, biopsy-proven acute cellular rejection (ACR) >1R, and chronic allograft vasculopathy (CAV) ≥1. Time to event analyses were performed using Cox proportional hazards models.
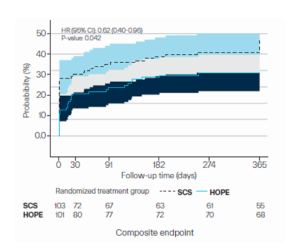
The early clinical benefits of HOPE observed at 30 days were sustained through the first year after transplantation. At 12 months, the composite endpoint occurred significantly less frequently in the HOPE group compared with the SCS group (32.7% vs 46.6%), corresponding to a hazard ratio of 0.62.
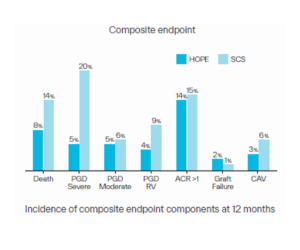
The most pronounced difference was observed for severe primary graft dysfunction, which was substantially reduced with HOPE. The high 92% survival observed in the HOPE group at 12 months was accompanied by a lower incidence of severe PGD, suggesting an association between improved early graft function and favourable clinical outcome. Rates of acute cellular rejection, graft failure, and cardiac allograft vasculopathy were comparable between groups.
European Heart Journal / 2026 /ehag264, doi.org/10.1093/eurheartj/ehag264
J Heart Lung Transplant, 2025 / DOI: doi.org/10.1016/j.healun.2025.09.020